38 Sr (Strontium)

Strontium is a soft, silver-yellow metal (Freshly cut strontium has a silvery appearance, but rapidly turns a yellowish color with the formation of the oxide).
it is similar to calcium and barium in physical and chemical properties.
Strontium is softer than calcium and decomposes in water more vigorously and quickly tarnishes in air.
It should be kept under kerosene to prevent from oxidation (out of contact with air and water)
It does not absorb nitrogen below 380°C.
Volatile strontium salts impart a beautiful crimson color to flames.
Natural strontium is a mixture of four stable isotopes.

Identity
CAS Number: CAS7440-24-6
CID Number: CID5359327
DOT Hazard Class: 4.2
DOT Number: 1383
CONTENT INDEX
Basic Properties of Strontium
Pronunciation: Stron-tee-am
Appearance: Silvery white Metallic (with a pale yellow tint)
Mass Number: 88
Standard Atomic weight: 87.62 g/mol
Atomic number (Z): 38
Electrons: 38
Protons: 38
Neutrons: 50
Period: 5
Group: 2
Block: s
Element category: Alkaline earth metal
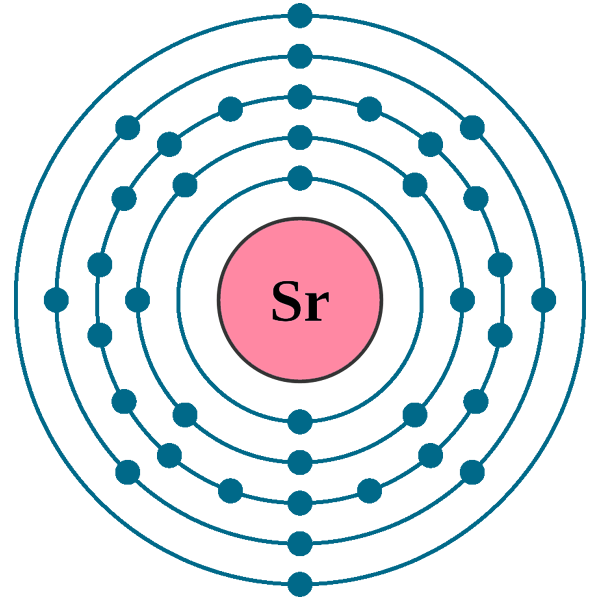
Electrons per shell: K2, L8, M18, N8, O2
Electron configuration: 1s22s22p63s23p63d104s24p65s2
Thermal Properties of Strontium
Phase: Solid
Melting point: 1050 K (777 oC, 1431 oF)
Boiling point: 1650 K (1377 oC, 2411 oF)
Debye temperature: 147 K (-126.15 oC, -195.07 oF)
Fusion heat: 7.43 kJ/mol
Vaporization heat: 141 kJ/mol
Specific heat: 300 J/(kg K)
Molar heat capacity: 26.4 J/(mol.K)
Thermal expansion: 22.5 μm/(m∙K)
Thermal conductivity: 35.4 W/(m∙K)
Electrical properties of Strontium
Electrical conductivity: 7.7×106 S/m
A Electrical resistivity: 132 nΩ∙m
A Electrical type: Conductor
Magnetic Properties of Strontium
A Magnetic type: Paramagnetic
Magnetic susceptibility (xmol): -92×10-6 cm3/mol
Volume magnetic susceptibility: 0.00000347
Mass magnetic susceptibility: 1.32×10-9 m3/kg
Molar magnetic susceptibility: 0.116×10-9 m3/mol
Physical Properties of Strontium
Density: 2.64 g/cm3 (In solid) 2.375 g/cm3 (In Liquid)
Molar volume: 0.000033316 m3/mol
Young’s modulus: 15.7 GPa
Shear modulus: 6.03 GPa
Mohs Hardness: 1.5
Poisson ratio: 0.28
Atomic Properties of Strontium
Oxidation states: 2,1,
Valence Electrons: 5s2
Ion charge: Sr2+
The ionization potential of an atom: 5.67
Ionization energies: 1st: 549.5 kJ.mol 2nd: 1064.2 kJ/mol 3rd: 4138 kJ/mol
Ionic radius: 112 pm (1.12 Å)
Atomic radius: empirical: 215 pm
Van der Waals: 249 pm
Covalent radius: 195±10 pm
Filling Orbital: 5s2
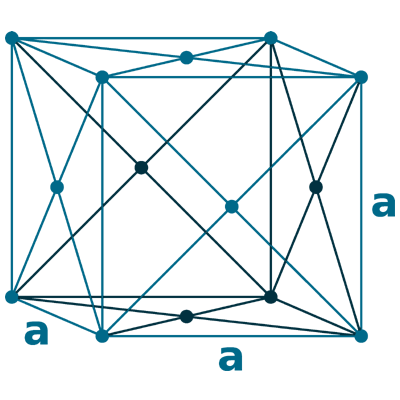
Crystal structure: Face-centered cubic
Lattice angles: π/2, π/2, π/2
Lattice constant: 608.49, 608.49, 608.49 pm
Grid parameters: a=6.080 Å
Space Group Name: Fm_3m
Space Group Number: 225
Reactivity of Strontium
Electronegativity: pauling scale: 0.95
Valence: +2
Electron affinity: 5.03 kJ/mol
Nuclear Properties of Strontium
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 1S0
Neutron cross section (Brans): 1.2
Neutron Mass Absorption: 0.0005
Isotopes: 82Sr 83Sr 84Sr 85Sr 86Sr 87Sr 88Sr 89Sr 90Sr
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 82Sr | Syn | – | 25.36 d |
| 83Sr | Syn | – | 1.35 d |
| 84Sr | 0.56 | 83.913 | Stable |
| 85Sr | Syn | – | 64.84 |
| 86Sr | 9.86 | 85.909 | Stable |
| 87Sr | 7 | 86.909 | Stable |
| 88Sr | 82.58 | 87.906 | Stable |
| 89Sr | Syn | – | 50.52 d |
| 90Sr | Trace | – | 28.90 y |
Chemical Reactions of Strontium
Strontium reacts with oxygen (O2)and forming a protective layer of SrO on the surface. When Ignited, Strontium metal burns in air and reacts with both oxygen and nitrogen, to give a mixture of white strontium oxide (SrO) and strontium nitride (Sr3N2)
2 Sr (s) + O2 (g) → 2 SrO (s)
3 Sr (s) + N2 (g) → 2 Sr3N2 (s)
Strontium reacts slowly with water and forming strontium hydroxide (Sr(OH)2)and hydrogen gas (H2).
Sr (s) + 2 H2O (l) → Sr(OH)2 (aq) + H2 (g)
Strontium react with Chlorine, bromine, iodine and forming Strontium (II) halides:
Sr (s) + Cl2 (g) → SrCl2 (s) (Strontium (ll) chloride)
Sr (s) + Br2 (g) → SrBr2 (s) (Strontium (ll) bromide)
Sr (s) + I2 (g) → SrI2 (s) (Strontium (ll) iodide)
The metal dissolves readily in dilute or concentrated hydrochloric acid (HCl) and forming Sr (II) ions and hydrogen gas.
Sr (s) + 2 HCl (aq) → Sr2+ (aq) + 2 Cl– (aq) + H2↑ (g)
Production:
The sulfate is reduced to the sulfide by the carbothermic reduction:
SrSO4 + 2 C → SrS + 2 CO2
The metal can also be prepared by electrolysis of a solution of strontium chloride in molten potassium chloride:
Sr2+ + 2 e− → Sr
2 Cl− → Cl2 + 2 e−
Strontium History
Naming: After the mineral strontianite, itself named after Strontian, Scotland
Discovery: William Cruickshank (1787)
First isolation: Humphry Davy (1808)
Strontium Uses
sStrontim has similar uses of calcium and a brium but it is employed rarely because of its higher cost.
Strontium compounds are used in pyrotechnics and gives the brilliant red colour to fireworks and flares.
It also has found use in producing ferrite magnets and in refining zinc.
It is one of the best high-energy beta-emitters.
Strontium-90 is a dangerously radioactive isotope, It is a by-product of nuclear reactors and present in nuclear fallout (from whose spent fuel is extracted).
It high-energy radiation can be used to generate electricity for space vehicles, remote weather stations and navigation buoys.
Modern ‘glow-in-the-dark’ paints and plastics contain strontium aluminate (SrAl2O4) , where they absorb light during the day and release it slowly for hours afterwards.
Strontium chloride hexahydrate (H12Cl2O6Sr) is an ingredient in toothpaste for sensitive teeth.
Strontium titanate (SrTiO3) is an optical material, which has extremely high refractive index (measure of the bending of a ray of light when passing from one medium into another) and an optical dispersion greater than that of diamond.
Biological role
It is Non-Toxic, Because it is similar to calcium.
In the human body, most of the absorbed strontium is deposited in the bones, In the ratio of strontium to calcium is between 1:1000 and 1:2000 roughly.
Radioactive strontium-90 is produced in nuclear explosions and released during nuclear plant accidents, which is particularly dangerous because it can be absorbed into the bones of young children.
For children exceeded strontium uptake may be a health risk, because it can cause problems with bone growth.
Abundance of Strontium
It is found chiefly in the minerals celestite and strontianite.
The metal can be prepared by electrolysis of the molten strontium chloride (SrCl2) mixed with potassium chloride (KCl), or by reducing strontium oxide (SrO) with aluminium in a vacuum at a temperature at which strontium distills off.
Three allotropic forms of the metal exist, with transition points at 235 and 540°C.
Annual world wide production is around 3,00,000 tons.
40×10-7% (In Universe)
86×10-5% (In Meteorites)
50×10-7% (In Sun)
0.036% (In Earth’s Crust)
0.0008% (In Oceans)
0.00046% (In Humans)


World’s Top 3 producers of Strontium
1) China
2) Spain
3) Mexico
World’s Top Reserve holders of Strontium
1) China
#Strontium


