75 Re (Rhenium)

It is a silvery white metal with a metallic luster, it is rarely seen as such on account of its high melting point, which is the 3rd highest after tungsten (3422 oC) & carbon (3550 oC).
Rhenium is very hard, and it resists corrosion but slowly tarnishes in moist air, and the annealed rhenium is very ductile, and can be bent, rolled, or coiled.

Identity
CAS Number: CAS7440-15-5
CID Number: CID23947
RTECS Number: RTECSVI0780000
CONTENT INDEX
Basic Properties of Rhenium
Pronunciation: Ree-nee-am
Appearance: Silvery grayish
Mass Number: 186
Standard Atomic weight: 186.207 g/mol
Atomic number (Z): 75
Electrons: 75
Protons: 75
Neutrons: 111
Period: 6
Group: 7
Block: d
Element category: Transition metal
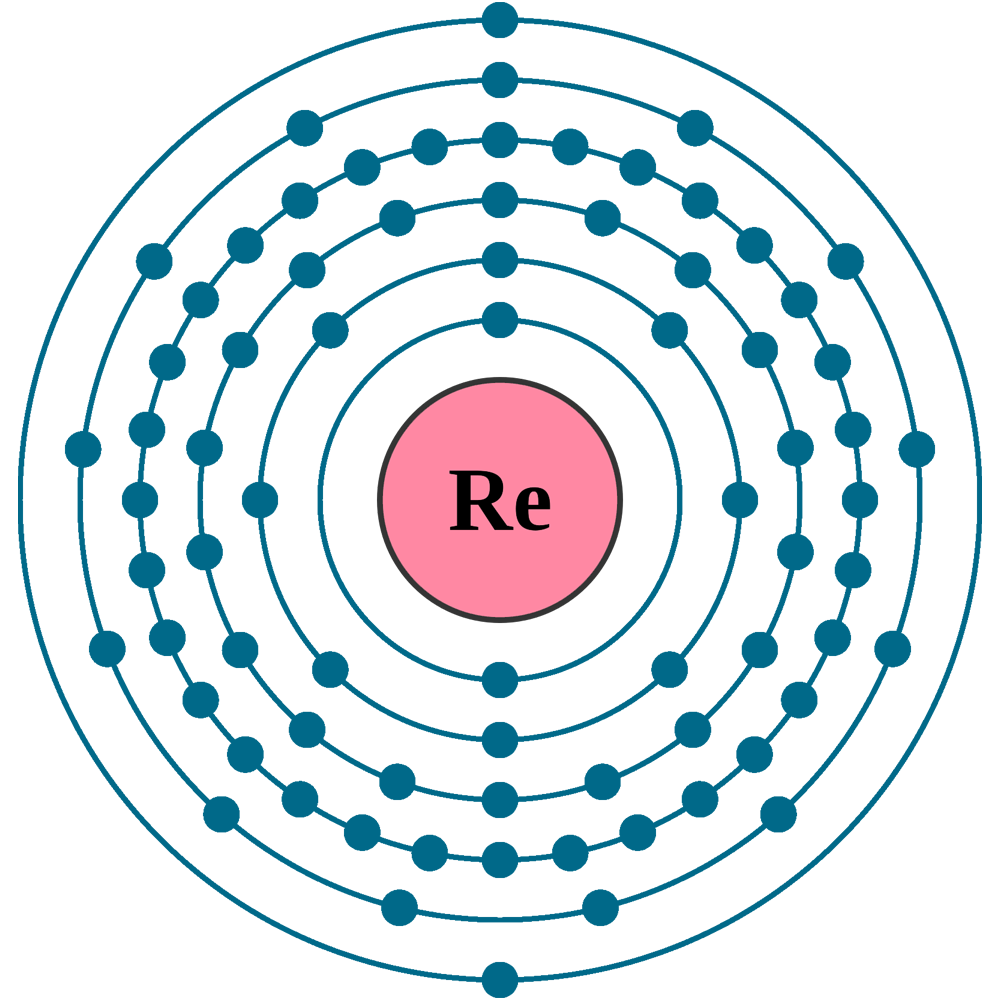
Electrons per shell: K2, L8, M18, N32, O13, P2
Electron configuration: 1s22s22p63s23p63d104s24p64d105s25p64f145d56s2
Thermal Properties of Rhenium
Phase: Solid
Melting point: 3459 K (3186 oC, 5767 oF)
Boiling point: 5903 K (5630 oC, 10,170 oF)
Debye temperature: 416 K (142.85 oC, 289.13 oF)
Fusion heat: 60.43 kJ/mol
Vaporization heat: 704 kJ/mol
Specific heat: 137 J/(kg K)
Molar heat capacity: 25.48 J/(mol.K)
Thermal expansion: 6.2 μm/(m∙K)
Thermal conductivity: 48 W/(m∙K)
Electrical properties of Rhenium
Electrical conductivity: 48×106 S/m
A Electrical resistivity: 193 nΩ∙m
A Electrical type: Conductor
Critical point (Superconducting point): 1.7 K (-271 oC, -456.61 o F)
Magnetic Properties of Rhenium
A Magnetic type: Paramagnetic
Magnetic susceptibility (xmol): +67.6×10-6 cm3/mol
Volume magnetic susceptibility: 0.0000959
Mass magnetic susceptibility: 4.56×10-9 m3/kg
Molar magnetic susceptibility: 0.849×10-9 m3/mol
Physical Properties of Rhenium
Density: 21.02 g/cm3 (In solid) 18.9 g/cm3 (In Liquid at M.P)
Molar volume: 0.0000088585 m3/mol
Young’s modulus: 463 GPa
Shear modulus: 178 GPa
Mohs Hardness: 7.0
Bulk modulus: 370 GPa
Poisson ratio: 0.30
Vicker hardness: 1350-7850 MPa
Brinell hardness: 1320-2500 MPa
Sound Speed: 4700 m/s
Atomic Properties of Rhenium
Oxidation states: -3, -1, 0, 1, 2, 3, 4, 5, 6, 7
Valence Electrons: 5d5 6s2
Ion charge: Re7+
Ionization energies: 1st: 760 kJ.mol 2nd: 1260 kJ/mol 3rd: 2510 kJ/mol
Ionic radius: 56 pm
Atomic radius: 137 pm (empirical)
Van der Waals: 217 Pm
Covalent radius: 151±7 pm
Filling Orbital: 5d5
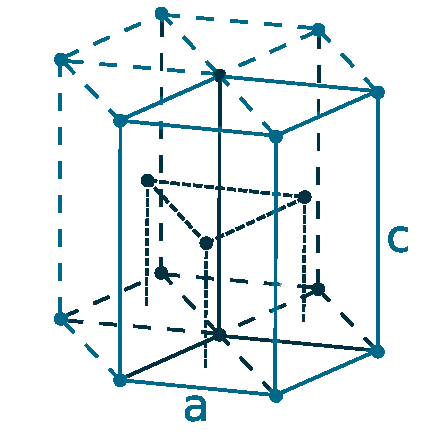
Crystal structure: Hexagonal close packed
Lattice angles: π/2, π/2, 2π/3
Lattice constant: 276.1, 276.1, 445.6 pm
Grid parameters: a=2.761 Å c=4.456 Å
Attitude c/a: 1.614
Space Group Name: P63/mmc
Space Group Number: 194
Reactivity of Rhenium
Electronegativity: 1.9 (pauling scale)
Valence: +7
Electron affinity: 14.5 kJ/mol
Nuclear Properties of Rhenium
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 6S5/2
Neutron cross section (Brans): 90
Neutron Mass Absorption: 0.016
Isotopes: 185Re 187Re
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 185Re | 37.4 | 184.952 | Stable |
| 187Re | 62.6 | 186.955 | 4.12×1010 y |
Chemical Reactions of Rhenium
It reacts and slowly tarnish in air under normal condition, if heated, it is oxidized to form Re (VII) oxide:
4 Re (s) + 7 O2 (g) → 2 Re2O7 (s)
Reacts with water, but not in normal condition:
Re (s) + 2 H2O (l) ⇌ ReO2 (s) + 4 H+ (aq) + 4 e– Eo = -0.251 V
Re3+ (aq) + 3 H2O (l) ⇌ ReO3 (s) + 6 H+ (aq) + 3 e– Eo = -0.318 V
Re3+ (aq) + 4 H2O (l) ⇌ ReO42- (aq) + 8 H+ (aq) + 3 e– Eo = -0.795 V
Re3+ (aq) + 4 H2O (l) ⇌ ReO4– (aq) + 8 H+ (aq) + 4 e– Eo = -0.422 V
Reacts with fluorine, when heated, and forming a mixture of Rhenium (VI) fluoride & rhenium (VII) fluoride.
Re (s) + 3 F2 (g) → ReF6 (s)
But at 400 oC & under pressure, only rhenium (VII) fluoride is formed.
2 Re (s) + 7 F2 (g) → 2 ReF7 (s)
The metal can dissolve in warm Bromine water.
The metal doesn’t dissolve in hydrochloric acid (HCl) & hydrofluoric acid (HF), but dissolve in nitric acid (HNO3) & concentrated sulphuric acid (H2SO4), and forming a solution of perrhenic acid (HReO4).
Rhenium History
Naming: After the river Rhine (German: Rhein)
Named by: Walter Noddack, Ida Noddack, Otto Berg (1925)
Discovery: Masataka Ogawa (1908)
First isolation: Masataka Ogawa (1919)
Rhenium Uses
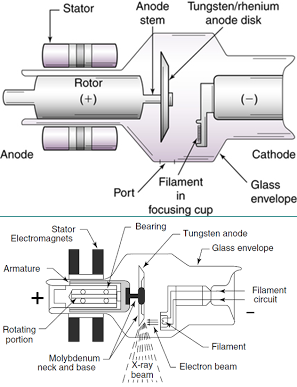
Rhenium is used as an additive to molybdenum and tungsten based alloys to give useful properties, where these alloys are used for X-ray machines(In rotating anode X-ray tube) & oven filaments.
It is also used as an electrical contact material, because of good wear and corrosion resistant property.
Molybdenum- Rhenium alloys are superconductive at 10 K (-263.15 oC).
Tungsten-Rhenium alloy are used to make Thermocouples for measuring temperatures up to 2200 oC, and used as a rhenium wire in photoflash lamps for photography.
It is an excellent catalyst for isomerization and hydrogenation.
platinum–rhenium alloy is used as catalysts for catalytic reforming (chemical process for making lead-free, high-octane gasoline).
Rhenium catalysts are extremely resistant to chemical poisoning (deactivation) from sulfur, nitrogen, & phosphorus, and so are used in certain kind of hydrogenation reactions for fine chemicals.
Rhenium is also added in nickel based superalloys for improved creep strength, which is used to make single-crystal turbine blades.
Biological role of Rhenium
Very little is known about its toxicity because it is used in very small amounts, but but two alloys potassium perrhenate (KO4Re) & rhenium trichloride (ReCl3) could have acute toxicity, and it should be handled with care until more data becomes available.
Abundance of Rhenium
Rhenium one of the rarest metal on Earth, where It does not occur uncombined in nature or as a compound in a distinct mineral species, however, it is widely spread throughout the earth’s crust to the extent of about 0.001 ppm (parts per million).
Commercially, rhenium is produced by extraction from the flue dust of molybdenum smelters (a process of applying heat to ore in order to extract out a base metal), which is obtained from copper-sulfide (Cu2S) ores.
Rhenium metal is prepared by reducing ammonium perrhentate (NH4ReO4) with hydrogen at elevated temperatures.
Commercial form of the element is usually powder, but it can be consolidated (combine into a single) by pressing and resistance-sintering in a vacuum or hydrogen atmosphere.
This process produces a compact shape in excess of 90% of the density of the metal.
Annual world wide production is around 50-60 tons.
2×10-8% (In Universe)
4.9×10-6% (In Meteorites)
1×10-8% (In Sun)
2.6×10-7% (In Earth’s Crust)
1×10-10% (In Oceans)
World’s Top 3 producers of Rhenium
1) Chile
2) USA
3) Poland
World’s Top 3 Reserve holders of Rhenium
1) Chile
2) USA
3) Russia
Rhenium Price: Pure (99.99%) metal price is around $2500-$3000 per KG (KiloGram)
#Rhenium


