61 Pm (Promethium)

Promethium is a soft beta emitter rare-earth metal.
X-radiation can be generated when beta particles impinge on elements of a high atomic number.
Promethium salts have a pink or red that glow in the dark with a pale blue or greenish, due to their high radioactivity.
It is very rare, so its chemical and physical properties are not well defined.


Identity
CAS Number: CAS7440-12-2
CONTENT INDEX
Basic Properties of Promethium
Pronunciation: Proh-mee-thee-am
Appearance: Metallic
Mass Number: 145
Standard Atomic weight: 146.915 g/mol
Atomic number (Z): 61
Electrons: 61
Protons: 61
Neutrons: 84
Period: 6
Block: f
Element category: Lanthanide
Electrons per shell: K2, L8, M18, N23, O8, P2
Electron configuration: 1s22s22p63s23p63d104s24p64d105s25p64f56s2
Thermal Properties of Promethium
Phase: Solid
Melting point: 1315 K (1042 oC, 1908 oF)
Boiling point: 3273 K (1900 oC, 5432 oF)
Fusion heat: 7.13 kJ/mol
Vaporization heat: 289 kJ/mol
Molar heat capacity: 24.3 J/(mol.K)
Thermal expansion: 9.0 μm/(m∙K)
Thermal conductivity: 17.9 W/(m∙K)
Electrical properties of Promethium
Electrical conductivity: 1.3×106 S/m
a Electrical resistivity: est. 0.75 μΩ∙m
a Electrical type: Conductor
Magnetic Properties of Promethium
Magnetic type: Paramagnetic
Physical Properties of Promethium
Density: 7.26 g/cm3
Molar volume: 0.00001996 m3/mol
Young’s modulus: α form: est. 46 GPa
Shear modulus: α form: est. 18 GPa
Bulk modulus: α form: est. 33 GPa
Poisson ratio: α form: est. 0.28
Vicker hardness: 410-440 MPa
Brinell hardness: 440-600 MPa
Sound Speed: 2130 m/s
Atomic Properties of Promethium
Oxidation states: 3,2
Valence Electrons: 4f5 6s2
Ion charge: Pm3+
Ionization energies: 1st: 540 kJ.mol 2nd: 1050 kJ/mol 3rd: 2150 kJ/mol
Ionic radius: 97.9 pm
Atomic radius: 236 pm (Van der Waals)
Covalent radius: 199 pm
Filling Orbital: 4f5
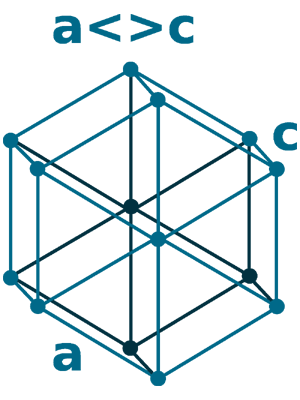
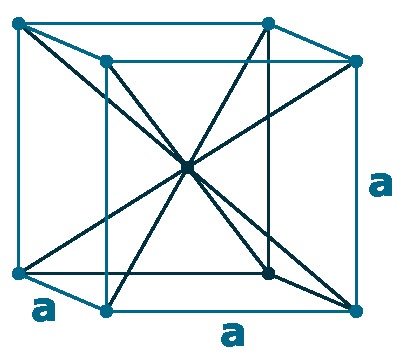
Crystal structure: Double Hexagonal close packed (change to Body centered cubic above 890 oC)
Grid parameters: a=3.65 Å c=11.65 Å
Attitude c/a: 3.19
Reactivity of Promethium
Electronegativity: pauling scale: 1.13
Valence: +3
Electron affinity: 50 kJ/mol
Nuclear Properties of Promethium
Half Life: 17.725 y
Lifetime: 25.558 y
Quantum Number: 6H5/2
Neutron cross section (Brans): 8000
Decay mode: Electron capture
Isotopes: 145Pm 146Pm 147Pm
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 145Pm | Trace | 144.913 | 17.7 y |
| 146Pm | Syn | – | 5.53 y |
| 147Pm | Trace | 146.915 | 2.623 y |
Promethium History
Discovery: Chien Shiung Wu, Emilio Segre, Hans Bethe (1942)
Discovery Location: Oak Ridge Tennennee United States
First isolation: Charles D. Coryell, Jacob A. Marinsky, Lawrence E. Glendenin, Harold G. Richter (1945)
Naming by: Grace Mary Coryell (1945)
Promethium Uses
Most promethium is used only for research purposes, Except for promethium-147
Promethium-147 have a useful life of about 5 years, which can be used as Beta radiation source for thickness measurement devices, and it can be absorbed by a phosphor to produce luminous light.
It is also used in specialized miniature (roughly the size of a drawing pin) Nuclear-powered batteries (capturing light in photocells which convert it into electric current) for guided missiles, watches, pacemakers, radios, and as a light source for signs or signals.
It is possible that in future promethium will be used as a X-ray source, and it may become useful as a heat source to provide auxiliary power for space probes and satellites.
Abundance of Promethium
Promethium-145 is the longest lived isotope, and has a specific activity of 940 Ci/g.
Longest-lived isotope of Promethium has a half-life is only 18 years. Due to this reason, it is not found naturally on Earth, It is completely missing from the earth’s crust. However, It has been identified in the spectrum of the star HR465 in Andromeda Galaxy.
A very small amounts of Promethiums can be produced by irradiating neodymium and praseodymium with neutrons, deuterons and alpha particles.
It also prepared from nuclear reactors, by ion exchange of nuclear reactor fuel processing wastes.
All commercially used promethium comes from nuclear reactors.
#promethium


