28 Ni (Nickel Element)

ANickel is silvery white, hard, malleable, ductile metal, which is somewhat ferromagnetic, and a fair conductor of heat and electricity.
It is of the iron-cobalt group and it takes on a high polish.
In its familiar compounds nickel is bivalent (molecule has a valence of two), although it assumes other valences.
Most nickel compounds are blue or green.
ANickel dissolves slowly in dilute acids but, like iron, becomes passive when treated with nitric acid.
Finely divided nickel adsorbs hydrogen.

Identity
CAS Number: CAS7440-02-0
CID Number: CID935
DOT Hazard Class: 4.1
DOT Number: 3089
RTECS Number: RTECSQR5950000
CONTENT INDEX
Basic Properties of Nickel
Appearance: Lustrous, Metallic, and silver with a gold tinge
Mass Number: 59
Standard Atomic weight: 58.70 g/mol
Atomic number (Z): 28
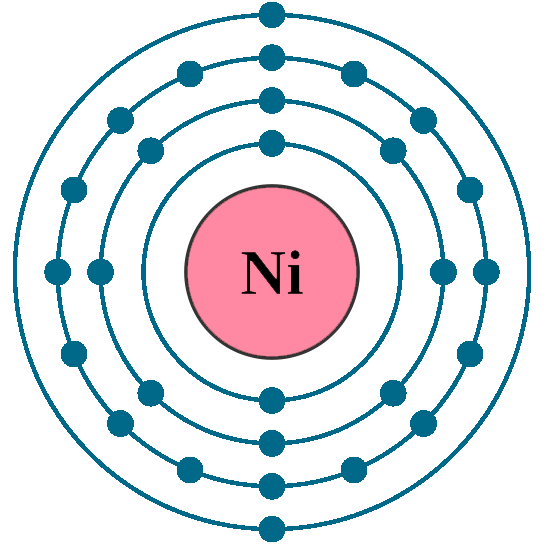
Electrons: 28
Protons: 28
Neutrons: 31
Period: 4
Group: 10
Block: d
Element category: Transition metal
Electrons per shell: K2, L8, M16, N2 OR K2, L8, M17, N1
Electron configuration: 1s22s22p63s23p63d84s2 OR 1s22s22p63s23p63d94s1
Thermal Properties of Nickel
Phase: Solid
Melting point: 1728 K (1455 oC, 2651 oF)
Boiling point: 3003 K (2730 oC, 4946 oF)
Debye temperature: 375 K (101.85 oC, 215.33 oF)
Fusion heat: 17.48 kJ/mol
Vaporization heat: 379 kJ/mol
Specific heat: 445 J/(kg K)
Molar heat capacity: 26.07 J/(mol.K)
Thermal expansion: 13.4 μm/(m∙K)
Thermal conductivity: 90.9 W/(m∙K)
Electrical properties of Nickel
Electrical conductivity: 14 x106 S/m
A Electrical resistivity: 69.3 nΩ∙m
A Electrical type: Conductor
Magnetic Properties of Nickel
A Magnetic type: Ferromagnetic
Curie point: 631 K (357.85 oC, 676.13 oF) (above which Ferromagnetism vanishes)
Magnetic susceptibility (xmol): +1860×10-6 cm3/mol
Physical Properties of Nickel
Density: 8.908 g/cm3 (In solid) 7.81 g/cm3 (In Liquid at M.P)
Molar volume: 0.000006588 m3/mol
Young’s modulus: 200 GPa
Shear modulus: 76 GPa
Mohs Hardness: 4.0
Bulk modulus: 180 GPa
Poisson ratio: 0.31
Vicker hardness: 638 MPa
Brinell hardness: 667-1600 MPa
Sound Speed: 4900 m/s
Atomic Properties of Nickel
Oxidation states: 4, 3, 2, 1, -1, -2
Valence Electrons: 3d8 4s2
Ion charge: Ni2+ Ni3+
The ionization potential of an atom: 7.61
Ionization energies: 1st: 737.1 kJ.mol 2nd: 1753 kJ/mol 3rd: 3395 kJ/mol
Ionic radius: 69 pm
Filling orbital: 3d8
Atomic radius: empirical: 124 pm
Van der Waals: 163 Pm
Covalent radius: 124±4 pm
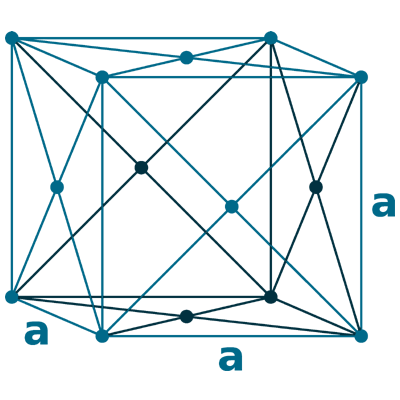
Crystal structure: Face centered cubic
Lattice angles: π/2, π/2, π/2
Lattice constant: 352.4, 352.4, 352.4 pm
Grid parameters: a=3.524 Å
Space Group Name: Fm_3m
Space Group Number: 225
Reactivity of Nickel
Electronegativity: pauling scale: 1.91
Valence: +2
Electron affinity: 112 kJ/mol
Nuclear Properties of Nickel
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 3F4
Neutron cross section (Brans): 4.5
Neutron Mass Absorption: 0.0026
Isotopes: 58Ni 59Ni 60Ni 61Ni 62Ni 63Ni 64Ni
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 58Ni | 68.077 | 57.935 | Stable |
| 59Ni | Trace | – | 7.6×104 y |
| 60Ni | 26.223 | 59.931 | Stable |
| 61Ni | 1.140 | 60.931 | Stable |
| 62Ni | 3.635 | 61.928 | Stable |
| 63Ni | Syn | – | 100 y |
| 64Ni | 0.926 | 63.928 | Stable |
Chemical Reactions of Nickel
The metal does not react with Air under ambient conditions, Finely divided nickel can burn, and forming nickel(II) oxide, NiO.
2 Ni (s) + O2 (g) → 2 NiO (s)
The metal reacts slowly with Halogens, forming Nickel (ll) halides:
Ni (s) + Cl2 (g) → NiCl2 (s) [yellow] (Nickel (ll) chloride)
Ni (s) + Br2 (g) → NiBr2 (s) [yellow] (Nickel (ll) bromide)
Ni (s) + I2 (g) → NiI2 (s) [black] (Nickel (ll) iodide)
Nickel(II) can be oxidized to nickel(III) using Br2 under alkaline conditions:
2 Ni(OH)2 (s) + Br2 (aq) + 2 OH– (aq) → 2 Ni(OH)3 (s) + 2 Br– (aq)
The metal dissolves slowly in dilute sulphuric acid to form solutions containing the aquated Ni(II) ion and hydrogen (H2). In this solution Ni(II) is present as the complex ion [Ni(H2O)6]2+.
Ni (s) + H2SO4 (aq) → Ni2+ (aq) + SO42- (aq) + H2 (g)
The strongly oxidizing concentrated nitric acid, HNO3, reacts on th surface of iron and passivates the surface.
Nickel History
Naming: German: kupfernickel (Devil’s copper).
Discovery and First isolation: Axel Fredrik Cronstedt (1751) in Stockholm Sweden.
Nickel Uses
ANickel resists corrosion, so it is extensively used for making stainless steel and other corrosion-resistant alloys such as Invar(R), Inconel(R), Monel(R), and the Hastelloys(R).
Nichrome is an alloy of Nickel (78.4%) and Chromium (20%) with small amounts of manganese, silicon, and iron, It resist corrosion, even at red hot temperature, So it is used in toasters, heaters, electric ovens, gas turbines, rocket engines etc..
Permalloy(R) is a nickel–iron magnetic alloy (80% Ni, 20% Fe), it is very high magnetic permeability.
Tubing made of copper-nickel (63% Ni, 28-34%Cu, 2%Mg, 2.5%Fe) alloy is not only hard but can resist corrosion by sea water, So it is extensively used in making desalination plants for converting sea water into fresh water, and It is ideal for propeller shaft in boats and turbine blades.
Copper-nickel alloy is also known as constantan, Eureka, Advance & Ferry.
Nickel steel is used to make armour plate and burglar-proof vaults
ANickel is used in batteries (including rechargeable nickel-cadmium batteries and nickel-metal hydride batteries used in hybrid vehicles), Adding on glass to gives it a green colour.
It is also used to make coins, In 19 century, US, UK, Canada used coins of aNickel (99.9%), so these coins were known as Nickel, in 2012 these coins were replaced with Nickel-plated steel, Causing allergy problems for some people and public controversy, and Now Coins are still made of Nickel But in form of alloy.
Finely divided nickel is used as a catalyst for hydrogenating vegetable oils.
Nickel plating is also used in ceramics, in the Edison(R) storage battery, and in the manufacture of Alnico magnets.

Biological role of Nickel
Biological role of nickel is uncertain, where, It can affect the growth of plants and also to be essential for some species.
Some nickel compounds (Nickel sulfide) can cause cancer(carcinogenic) if the fume and dust is inhaled, and some people are allergic to contact with the metal.
ANickel cannot be avoided completely, we intake nickel compounds with our diet, Because It is an essential element for some beans, such as the navy bean that is used for baked beans.
Exposure to nickel metal and soluble compounds (as Ni) should not exceed 0.05 mg/cm3.
Abundance of Nickel
The minerals from which most nickel is extracted are iron/nickel sulfides such as pentlandite (Fe,Ni)9S8) and pyrrhotite Fe1−xS (x = 0 to 0.2).
Iron meteorites (Fe, Ni (5%-25%) & Co (>95%)), or siderites (FeCO3), may contain iron alloyed with from 5% to nearly 20% nickel.
Hundreds of millions of years ago, a substantial amount of the nickel on Earth arrived with meteorites, and the landed region was Ontario (Canada), where, commercially produces about 30 percent of the world’s supply of nickel.
Annual world wide production is around 2,000,000 tons.
0.006% (In Universe)
1.3% (In Meteorites)
0.008% (In Sun)
0.009% (In Earth’s Crust)
2×10-7% (In Oceans)
0.00001% (In Humans)


World’s Top 3 producers of Nickel
1) Russia
2) Indonesia
3) Philippines
World’s Top 3 Reserve holders of Nickel
1) Australia
2) New Caledonia
3) Brazil
#Nickel


