10 Ne (Neon Element)

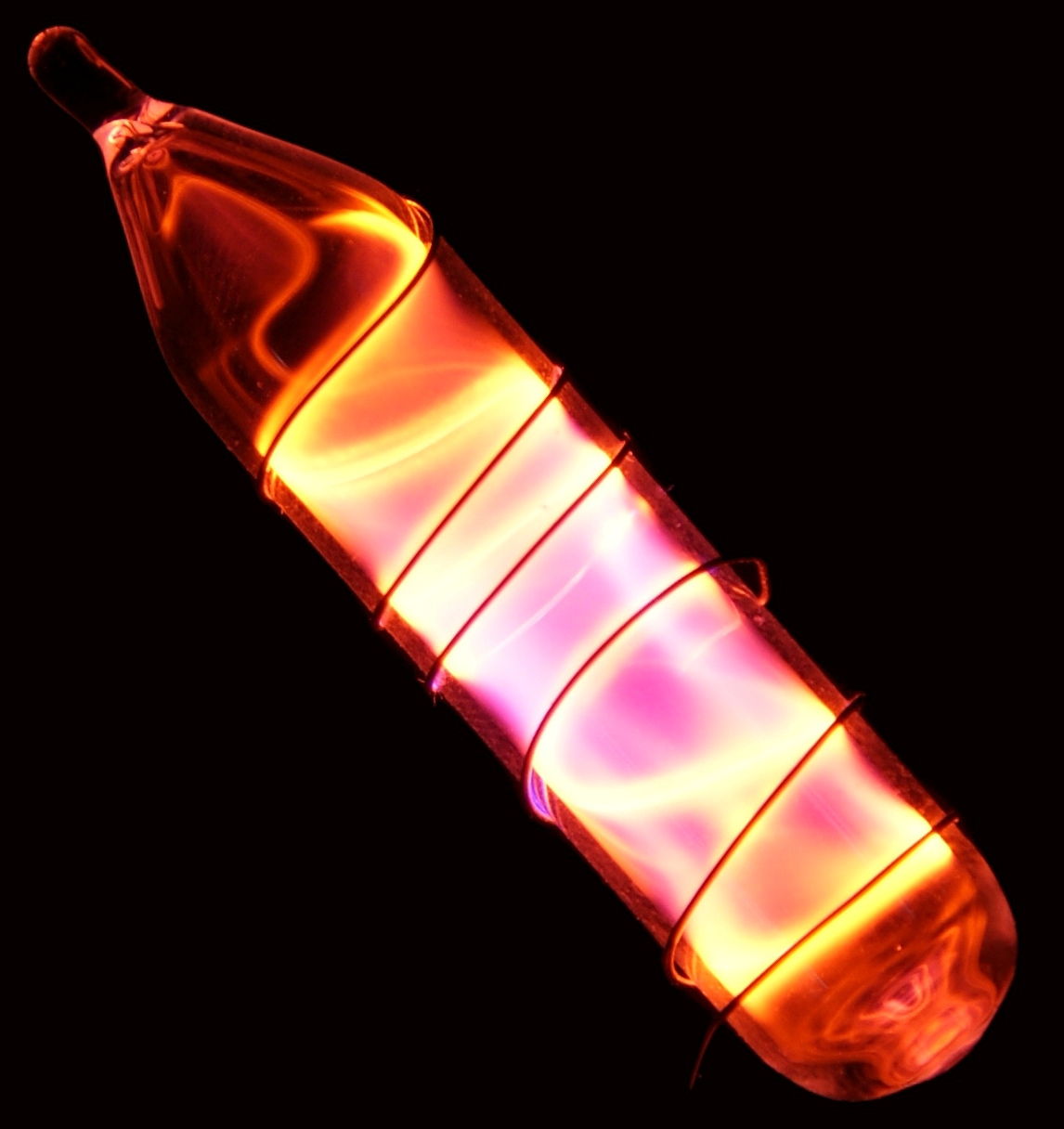
Neon is the 2nd lightest noble gas, its colour is reddish-orange glows in Neon lamps.
Refrigerating capacity of liquid Neon (per unit volume) is over 40 times of liquid helium and 3 times of liquid hydrogen, Even Neon is also less expensive refrigerant than helium in most applications.
All of the rare gases, the discharge of neon is the most intense at ordinary voltages and currents.
Neon is a very inert element, and it can form an exotic compound with fluorine.
If neon or true compounds of neon exist, then It is still questionable, but some evidence suggests that this may be true.
The ions, Ne+, (NeAr)+, (NeH)+, and (HeNe+) are have also been observed from optical and mass spectrometric studies.
Neon also forms an unstable hydrate.

Identity
CAS Number: CAS7440-01-9
CID Number: CID23935
DOT Hazard Class: 2.2
DOT Number: 1913
RTECS Number: RTECSQP4450000
CONTENT INDEX
Basic Properties of Neon
Appearance: Colour less but glow in orange-red when place in an electric field
Mass Number: 20
Standard Atomic weight: 20.179 g/mol
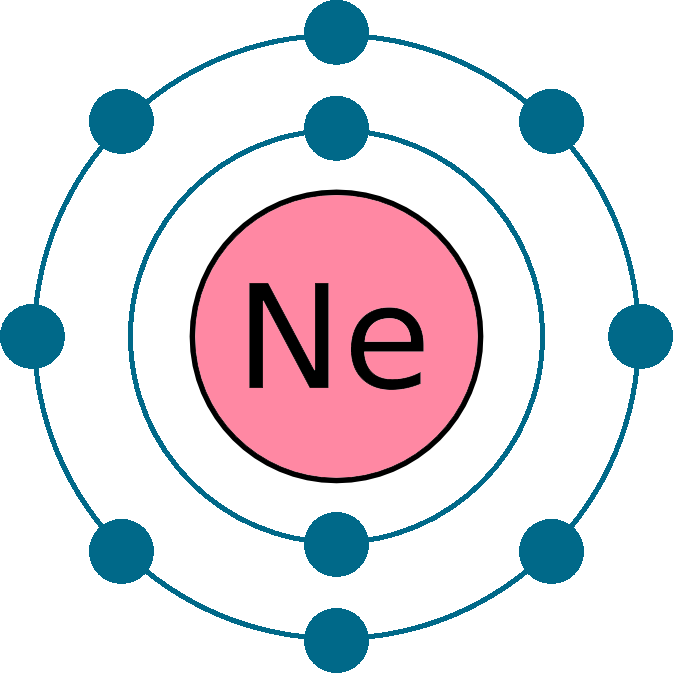
Atomic number (Z): 10
Electrons: 10
Protons: 10
Neutrons: 10
Period: 2
Group: 18
Block: p
Element category: Noble gas
Electrons per shell: K2, L8
Electron configuration: 1s22s22p6
Thermal Properties of Neon
Phase: Gas
Melting point: 24.55 K (-248.6 oC, -415.48 oF)
Boiling point: 27.10 K (-246.05 oC, -410.89 oF)
Debye temperature: 63.5 K (-209.65 oC, -345.37 oF)
Triple point Temperature: 24.55 K (-248.6 oC, -415.48 oF)
Triple point Pressure: 43.37 KPa (0.4280 Atm)
Critical point Temperature: 44.49 K (-228.66 oC, -379.588 oF)
Critical point Pressure: 2.768 MPa (27.318 Atm)
Fusion heat: 0.335 kJ/mol
Vaporization heat: 1.70 kJ/mol
Specific heat: 1030 J/(kg K)
Molar heat capacity: 20.78 J/(mol.K)
Thermal conductivity: 49.12×10-3 W/(m∙K)
Magnetic Properties of Neon
Magnetic type: Diamagnetic
Magnetic susceptibility (xmol): -6.75×10-6 cm3/mol
Volume magnetic susceptibility: 0.00081618
Mass magnetic susceptibility: -4.1×10-9 m3/kg
Molar magnetic susceptibility: -827×10-9 m3/mol
Physical Properties of Neon
Density: 0.9 g/L (In gas), 1.206 g/cm3 (In Liquid at B.P)
Molar volume: 0.02242 m3/mol
Sound Speed: 435 m/s
Atomic Properties of Neon
Oxidation states: 0
Valence Electrons: 2s2 2p6
Ionization potential of an atom: 21.46
Ionization energies: 1st: 2080.6 kJ.mol 2nd: 3952.4 kJ/mol 3rd: 6122 kJ/mol
Atomic radius: 38 pm (empirical)
Van der Waals: 154 Pm
Covalent radius: 58 pm
Filling Orbital: 2p6
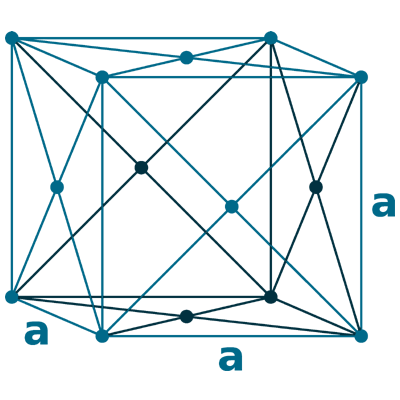
Crystal structure: Face centered cubic (F.C.C)
Lattice angles: π/2, π/2, 2π/3
Lattice constant: 362.1, 362.1, 2625 pm
Grid parameters: a=4.430 Å
Space Group Name: R_3m
Space Group Number: 166
Nuclear Properties of Neon
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 1S0
Neutron cross section (Brans): 0.04
Neutron Mass Absorption: 0.00059
Isotopes: 20Ne 21Ne 22Ne
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 20Ne | 90.47 | 19.991 | Stable |
| 21Ne | 0.28 | 20.993 | Stable |
| 22Ne | 9.25 | 21.992 | Stable |
Chemical Reactions of Neon
It Doesn’t reaction with Air, Water, Halogen, Acid, Bases etc..
Neon History
Naming: Greek: neos (new)
Prediction: William Ramsay (1898)
Discovery & First Isolation: William Ramsay & Morris Travers (1898)
Neon Uses
In a vacuum discharge tube, The reddish-orange color emitted in neon lights is widely used to make advertising signs.
Only the Red signs actually contain pure neon.

Neon is also used to make high-voltage indicators and switching gear, wave meter tubes, TV tubes, lightning arresters, and diving equipment.
Helium and Neon are used to make a type of gas laser.
Liquid neon is commercially used as an economical cryogenic refrigerant, which has over 40 times more refrigerating capacity per unit volume than liquid helium, and more than 3 times that of liquid hydrogen.
Biological role
It has Non-toxicity, But Liquid neon evaporates very quickly causing a serious risk of suffocation.
Effect of exposure: Simple Asphyxiant by Inhalation:, Frostbite: when skin contact with liquid, Frostbite: when eyes contact with Liquid.
Abundance of Neon
Neon is the 5th most abundant element in the universe, after Hydrogen, Helium, Oxygen and Carbon. However, it is present in the Earth’s atmosphere at a concentration of just 18.2 ppm (parts per million) or 1 part in 55,000.
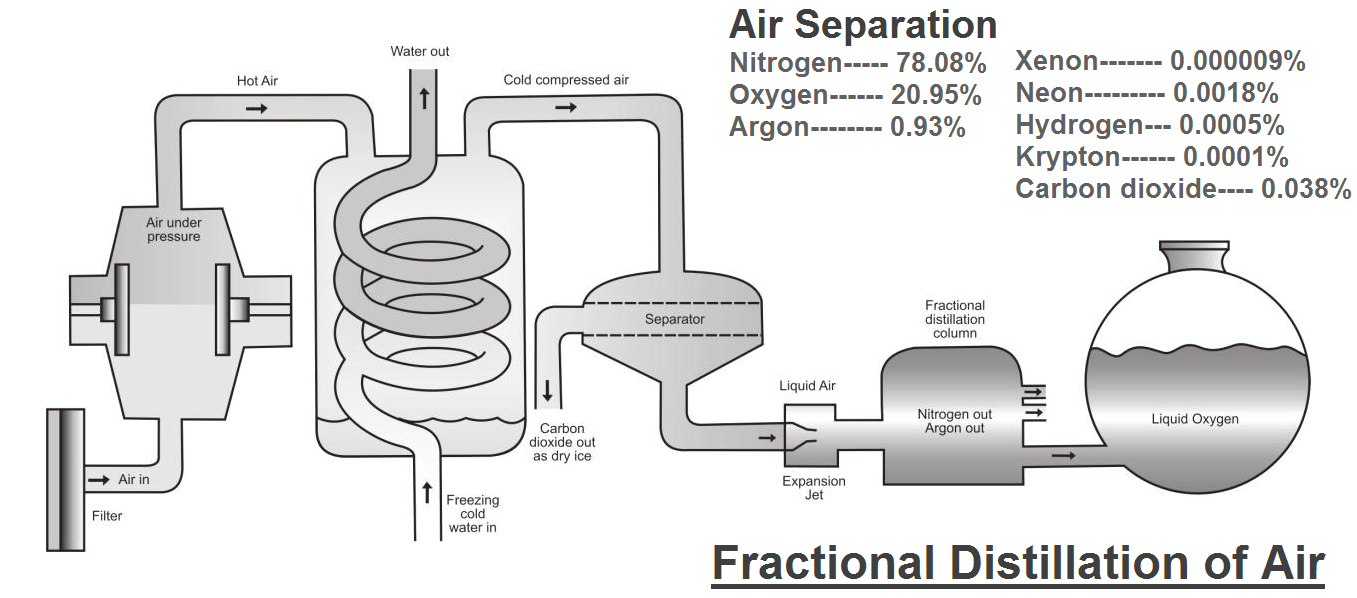
Neon is usually found in the form of a gas with molecules consisting of a single Neon atom and It is extracted by fractional distillation of liquid air. This gives a fraction that contains both helium and neon.
The helium (He) is removed from the mixture with activated charcoal.
0.13% (In Universe)
N/A (In Meteorites)
0.1% (In Sun)
3×10-7% (In Earth’s Crust)
1.2×10-8% (In Oceans)
Neon Price
Element price is around $320-$350per KG (KiloGram). Price of Extracted Liquid Neon can be more than 55 times costly that of liquid helium.
#Neon


