49 In (Indium)

It is available in ultra pure form
It is very soft, ductile, lustrous, silvery-white metal.
Indium is stable in air and water but dissolves in acids.
When Indium heated above its melting point it ignites burning with a violet flame.
Pure metal gives a high-pitched “cry” when bent.
Gallium and indium are able to wet glass.

Identity
CAS Number: CAS7440-74-6
CID Number: CID5359967
DOT Hazard Class: 4.1
DOT Number: 3089
RTECSNL Number: RTECSNL1050000
CONTENT INDEX
Basic Properties of Indium
Pronunciation: In-dee-am
Appearance: Silvery lustrous gray
Mass Number: 115
Standard Atomic weight: 114.818 g/mol
Atomic number (Z): 49
Electrons: 49
Protons: 49
Neutrons: 66
Period: 5
Group: 13
Block: p
Element category: Post-transition metal
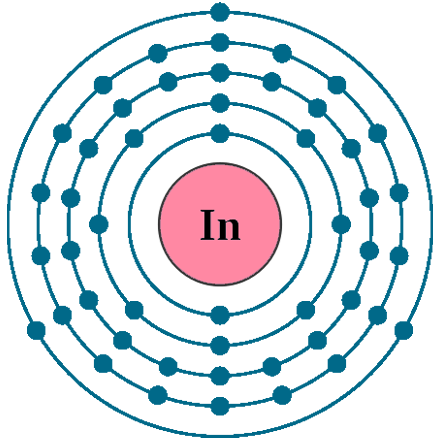
Electrons per shell: K2, L8, M18, N18, O3
Electron configuration: 1s22s22p63s23p63d104s24p64d105s25p1
Thermal Properties of Indium
Phase: Solid
Melting point: 429.7485 K (156.5985 oC, 313.8773 oF)
Boiling point: 2345 K (2072 oC, 3762 oF)
Debye temperature: 129 K (-144.15 oC, -227.47 oF)
Fusion heat: 3.281 kJ/mol
Vaporization heat: 231.8 kJ/mol
Molar heat capacity: 26.74 J/(mol.K)
Thermal expansion: 32.1 μm/(m∙K)
Thermal conductivity: 81.8 W/(m∙K)
Electrical properties of Indium
Electrical conductivity: 1.1×106 S/m
A Electrical resistivity: 83.7 nΩ∙m
A Electrical type: Conductor
Critical point (Superconducting point): 3.41 K
Magnetic Properties of Indium
A Magnetic type: Diamagnetic
Magnetic susceptibility (xmol): -64×10-6 cm3/mol
Volume magnetic susceptibility: 0.00081618
Mass magnetic susceptibility: 111×10-9 m3/kg
Molar magnetic susceptibility: 16.69×10-9 m3/mol
Physical Properties of Indium
Density: 7.52 g/cm3 (In solid) 7.16 g/cm3 (In Liquid)
Molar volume: 0.00001570 m3/mol
Young’s modulus: 11 GPa
Mohs Hardness: 1.2
Poisson ratio: 0.274
Brinell hardness: 8.8-10 MPa
Sound Speed: 1215 m/s
Atomic Properties of Indium
Oxidation states: 3, 2, 1, -1, -2, -5
Valence Electrons: 5s2 5p1
Ion charge: In3+
The ionization potential of an atom: 8.95
Ionization energies: 1st: 558.3 kJ.mol 2nd: 1820.7 kJ/mol 3rd: 2704 kJ/mol
Ionic radius: 80 pm
Van der Waals: 193 pm
Atomic radius: empirical: 167 pm
Covalent radius: 142±5 pm
Filling Orbital: 5p1
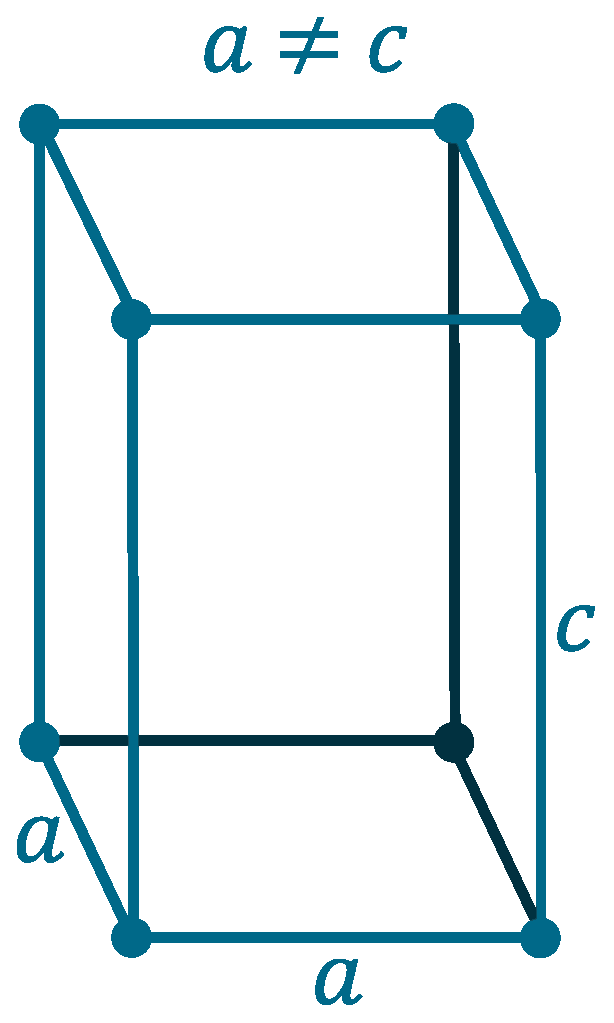
Crystal structure: Tetragonal
Lattice angles: π/2, π/2, π/2
Lattice constant: 3.252, 3.252, 4.946 pm
Grid parameters: a=3.252 Å c=4.946 Å
Attitude c/a: 1.52
Space Group Name: l4/mmm
Space Group Number: 139
Reactivity of Indium
Electronegativity: pauling scale: 1.17
Valence: +3
Electron affinity: 28.9 kJ/mol
Nuclear Properties of Indium
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 2P1/2
Neutron cross section (Brans): 194
Neutron Mass Absorption: 0.06
Isotopes: 113In 113In
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 113In | 4.28 | – | Stable |
| 115In | 95.72 | – | 4.41×1014 y |
Chemical Reactions
Indium reacts with water to reproduce soluble indium(III) hydroxide:
In(OH)3 + 3 HCl → InCl3 + 3 H2O
Indium History
Naming: From the indigo blue it shows in a spectroscope.
Discovery: Ferdinand Reich and Hieronymous Theodor Richter (1863) in Germany
First isolation: Hieronymous Theodor Richter (1863)
Indium Uses
Indium is found in low-melting fusible alloys (allow of 24% indium – 76% gallium is liquid at room temperature), It’s used in making bearing alloys, rectifiers, germanium transistors, fire-sprinkler systems in shops and warehouses, thermistors, and photoconductors.
A Indium tin oxide (ITO) is an important part of Flatscreen TVs, Touchscreens, and solar panels.
A Indium nitride, antimonide and phosphide are semiconductors used in transistors and microchips.
The Indium metal is plated to glass to form atmospheric corrosion-resistant mirror surface that can be used to give a mirror finish to windows of tall buildings, and as a protective film on welders’ goggles.
Indium foils are used to assess what is going on inside nuclear reactors.
Biological role: Indium has a low order of toxicity and if it is consumed more than a few milligrams that can affect the development of an embryo or foetus.
Abundance of Indium
Indium is one of the least abundant minerals on Earth, it is most frequently associated with zinc Ores. It is commercially produced as a by-product of zinc refining. However, it is also found in iron, lead, and copper ores.
Annual world wide production is around 75 tons.
3×10-8% (In Universe)
4.4×10-6% (In Meteorites)
4×10-7% (In Sun)
0.000016% (In Earth’s Crust)
1×10-11% (In Oceans)
World’s Top 3 producers of Indium
1) China
2) Republic of Korea
3) Japan
#indium


