66 Dy (Dysprosium)

It is a soft, metallic to bright silver luster, can be cut with knife and easily machined without sparking.
It is very stable in air, but it reacts readily with oxygen and water and dissolves in mineral acids.

Identity
CAS Number: CAS7429=91-6
CID Number: CID23912
CONTENT INDEX
Basic Properties of Dysprosium
Pronunciation: Dis-pro-zee-am
Appearance: Silvery white
Mass Number: 162
Standard Atomic weight: 162.500 g/mol
Atomic number (Z): 66
Electrons: 66
Protons: 66
Neutrons: 96
Period: 6
Block: f
Element category: Lanthanide
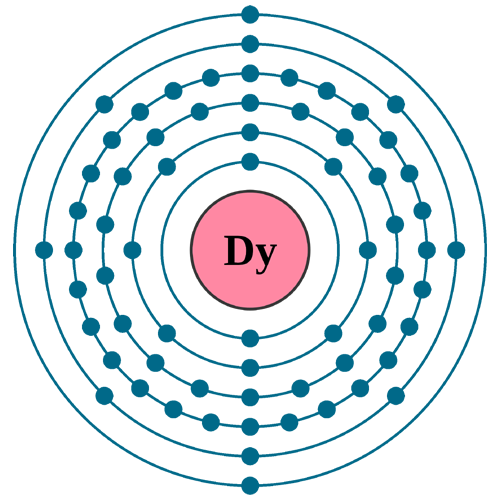
Electrons per shell: K2, L8, M18, N28, O8, P2
Electron configuration: 1s22s22p63s23p63d104s24p64d105s25p64f106s2
Thermal Properties of Dysprosium
Phase: Solid
Melting point: 1680 K (1407 oC, 2565 oF)
Boiling point: 2840 K (2562 oC, 4653 oF)
Fusion heat: 11.06 kJ/mol
Vaporization heat: 280 kJ/mol
Molar heat capacity: 27.7 J/(mol.K)
Thermal expansion: α, poly: 9.9 μm/(m∙K)
Thermal conductivity: 10.7 W/(m∙K)
Neel Point: 178 K
Electrical properties of Dysprosium
Electrical conductivity: 1.1 x106 S/m
s Electrical resistivity: α, poly: 926 μΩ∙m
s Electrical type: Conductor
Curie point: 87 K
Magnetic Properties of Dysprosium
s Magnetic type: Paramagnetic
Magnetic susceptibility (xmol): +103,500×10-6 cm3/mol
Volume magnetic susceptibility: 0.046604
Mass magnetic susceptibility: 5450×10-9 m3/kg
Molar magnetic susceptibility: 885.625×10-9 m3/mol
Physical Properties of Dysprosium
Density: 8.540 g/cm3 (In solid) 8.37 g/cm3 (In Liquid)
Molar volume: 0.000019 m3/mol
Young’s modulus: α form: 61.4 GPa
Shear modulus: α form: 24.7 GPa
Mohs Hardness: 1.65
Bulk modulus: α form: 40.5 GPa
Poisson ratio: α form: 0.247
Vicker hardness: 410-550 MPa
Brinell hardness: 500-1050 MPa
Sound Speed: 2710 m/s
Atomic Properties of Dysprosium
Oxidation states: 4, 3, 2, 1
Valence Electrons: 4f10 6s2
Ion charge: Dy3+
Ionization energies: 1st: 573.0 kJ.mol 2nd: 1130 kJ/mol 3rd: 2200 kJ/mol
Ionic radius: 91.2 pm
Atomic radius: 229 pm (Van der Waals)
Covalent radius: 192±7 pm
Filling Orbital: 4f10
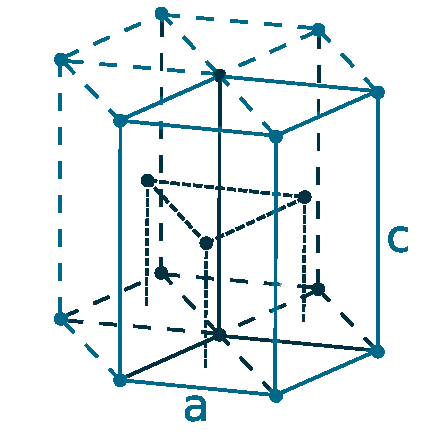
Crystal structure: Hexagonal close-packed
Lattice angles: π/2, π/2, π/3
Lattice constant: 359.3, 359.3, 565.39 pm
Grid parameters: a=3.593 Å, c=5.654 Å
Attitude c/a: 1.574
Space Group Name: P63/mmc
Space Group Number: 194
Reactivity of Dysprosium
Electronegativity: pauling scale: 1.22
Valence: +3
Electron affinity: 50 kJ/mol
Nuclear Properties of Dysprosium
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 5I8
Neutron cross section (Brans): 1010
Neutron Mass Absorption: 0.2
Isotopes: 154Dy 156Dy 158Dy 160Dy 161Dy 162Dy 163Dy 164Dy
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 154Dy | Syn | – | 3×106 y |
| 156Dy | 0.056 | 155.924 | Stable |
| 158Dy | 0.095 | 157.924 | Stable |
| 160Dy | 2.329 | 159.925 | Stable |
| 161Dy | 18.889 | 160.927 | Stable |
| 162Dy | 25.475 | 161.927 | Stable |
| 163Dy | 24.896 | 162.929 | Stable |
| 164Dy | 28.260 | 163.929 | Stable |
Chemical Reactions
Dysprosium metal tarnishes slowly in air and burns readily to form a dysprosium (lll) oxide:
4 Dy + 3 O2 → 2 Dy2O3
Reacts slowly with cold water and quickly with hot water (form dysprosium hydroxide and hydrogen gas):
2 Dy (s) + 6 H2O (l) → 2 Dy(OH)3 (aq) + 3 H2 (g)
The metal reacts with all Halogens (At above 200 oC) to form Dysprosium (lll) halides:
2 Dy (s) + 3 F2 (g) → 2 DyF3 (s) [green] (Dysprosium (lll) fluoride)
2 Dy (s) + 3 Cl2 (g) → 2 DyCl3 (s) [white] (Dysprosium (lll) chloride)
2 Dy (s) + 3 Br2 (g) → 2 DyBr3 (s) [white] (Dysprosium (lll) bromide)
2 Dy (s) + 3 I2 (g) → 2 DyI3 (s) [green] (Dysprosium (lll) iodide)
Dissolves readily in dilute sulfuric acid to form Solutions containing Dysprosium (lll) ions (yellow):
2 Dy (s) + 3 H2SO4 (aq) → 2 Dy3+ (aq) + 3 SO42− (aq) + 3 H2 (g)
Dysprosium fluoride, or dysprosium chloride can be reduced by using either Calcium or Lithium.
3 Ca + 2 DyF3 → 2 Dy + 3 CaF2
3 Li + DyCl3 → Dy + 3 LiCl
Dysprosium History
Naming: From Holmia (Greek name of Sweden)
Discovery: Lecoq de Boisbaudran
Dysprosium Uses
It is used in alloys with special stainless steels for neodymium-based magnets, Because it can resist to demagnetize at high temperature.
These magnets are used in wind turbines, electrical vehicles etc., so demand of dysprosiums is growing rapidly.
s
Dysprosium oxide-nickel cement (composition of ceramic and metal) used in cooling nuclear reactor control rods.
This cement can readily absorbs neutrons without swelling or contracting When neutrons bombarded for long periods.
s
Dysprosiums is also used in making laser materials, By combination with vanadium and other rare earths.
A
Dysprosium iodide is best known use in halide discharge lamps to get very intense white light.
Dysprosium-cadmium chalcogenides is a sources of infrared radiation, which is used for studying chemical reactions.
Biological role: It is Low-toxic, But it should be handled with care.
Abundance of Dysprosium
Like many other Lanthanides, It is found in the minerals monazite and bastnaesite.
It can also found in smaller quantities in some other minerals such as fergusonite and xenotime.
Dysprosium can be extracted by ion exchange and solvent extraction.
It can also be prepared by the reduction of dysprosium trifluoride with calcium metal
Annual world production is around 100 tons.
2×10-7% (In Universe)
2.7×10-5% (In Meteorites)
2×10-7% (In Sun)
0.00062% (In Earth’s Crust)
9.1×10-11% (In Oceans)
World’s Top 3 producers of Dysprosium
1) China
2) Russia
3) Malaysia
World’s Top 3 Reserve holders of Dysprosium
1) China
2) CIS Countries (inc. Russia)
3) USA
#Dysprosium


