13 Al (Aluminium Element)

Pure aluminum is a Light-weight, Nonmagnetic, Nonsparking, silvery-white metal, It is the 2nd most malleable and the 6th most ductile metal.
It forms thin layer of oxidation quickly, when it is exposed to air.
Aluminum has only one naturally occurring isotope (27Al), which is not radioactive.

Identity
CAS Number: CAS7429-90-5
CID Number: CID5359268
DOT Hazard Class: 4.3
DOT Number: 1396
RTECS Number: RTECSBD0330000
CONTENT INDEX
Basic Properties of Aluminium
Alternative name: Aluminum
Pronunciation: AL-yuu-min-ee-am / A-Lew-min-am
Appearance: Silvery gray Metallic
Mass Number: 27
Standard Atomic weight: 26.981 g/mol
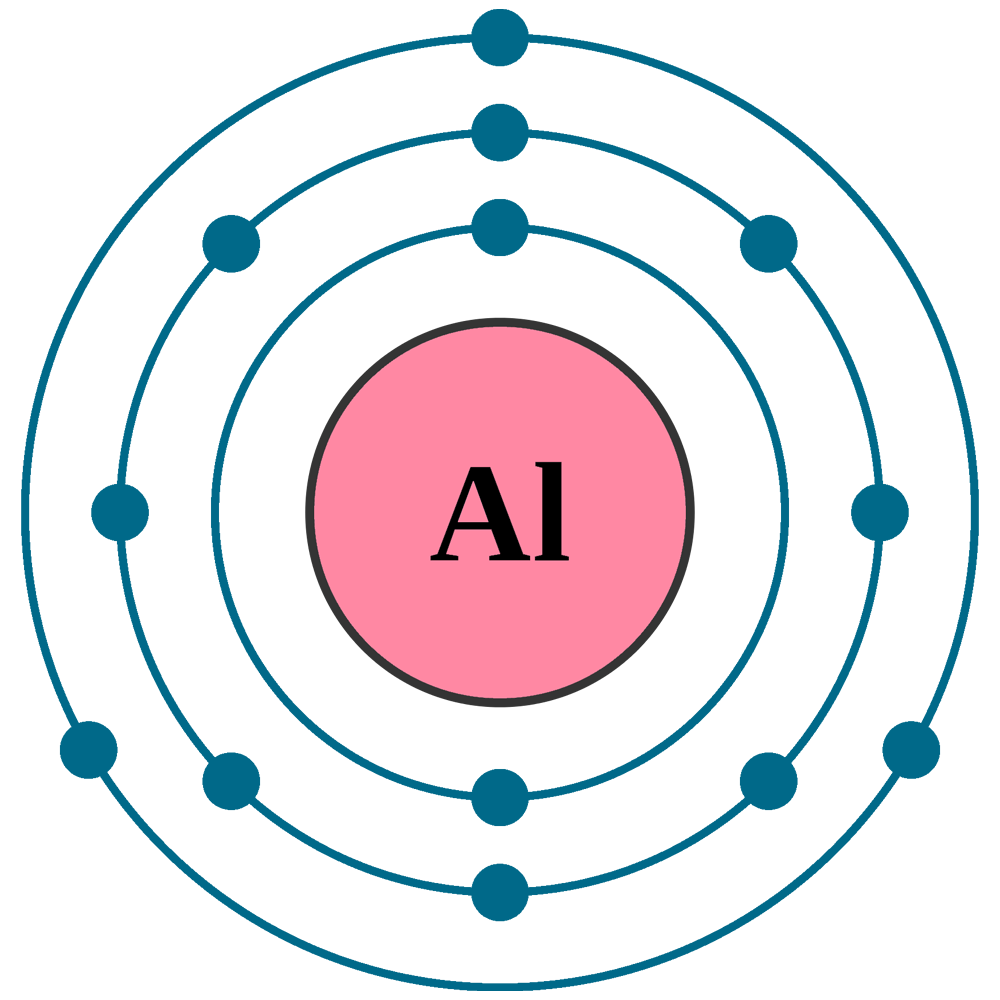
Atomic number (Z): 13
Electrons: 13
Protons: 13
Neutrons: 14
Period: 3
Group: 13
Block: p
Element category: Post Transition metal
Electrons per shell: K2, L8, M3
Electron configuration: 1s22s22p63s23p1
Thermal Properties of Aluminium
Phase: Solid
Melting point: 933.5 K (660.35 oC, 1220.63 oF)
Boiling point: 2743 K (2469.85 oC, 4477.73 oF)
Debye temperature: 394 K (120.85 oC, 249.53 oF)
Fusion heat: 10.71 kJ/mol
Vaporization heat: 284 kJ/mol
Specific heat: 904 J/(kg K)
Molar heat capacity: 24.20 J/(mol.K)
Thermal expansion: 23.1 μm/(m∙K)
Thermal conductivity: 237 W/(m∙K)
Electrical properties of Aluminium
Electrical conductivity: 38×106 S/m
A Electrical resistivity: 26.5 nΩ∙m
A Electrical type: Conductor
Critical point (Superconducting point): 1.18 K (-271.97 oC, -457.55 oF)
Magnetic Properties of Aluminium
A Magnetic type: Paramagnetic
Magnetic susceptibility (xmol): +16.5×10-6 cm3/mol
Volume magnetic susceptibility: 0.0000211
Mass magnetic susceptibility: 7.8×10-9 m3/kg
Molar magnetic susceptibility: 0.21×10-9 m3/mol
Physical Properties of Aluminium
Density: 2.70 g/cm3 (In solid) 2.37 g/cm3 (In Liquid at M.P)
Molar volume: 0.00000999 m3/mol
Young’s modulus: 70 GPa
Shear modulus: 26 GPa
Mohs Hardness: 2.75
Bulk modulus: 76 GPa
Poisson ratio: 0.35
Vicker hardness: 160-350 MPa
Brinell hardness: 160-550 MPa
Sound Speed: 5000 m/s
Atomic Properties of Aluminium
Oxidation states: +3, +2, +1, -1, -2
Valence Electrons: 3s2 3p1
Ion charge: Al3+
Ionization potential of an atom: 5.95
Ionization energies: 1st: 577.5 kJ.mol 2nd: 1816.5 kJ/mol 3rd: 2745 kJ/mol
Ionic radius: 0.535 pm
Atomic radius: 143 pm (empirical)
Van der Waals: 184 Pm
Covalent radius: 121±4 pm
Filling Orbital: 3p1
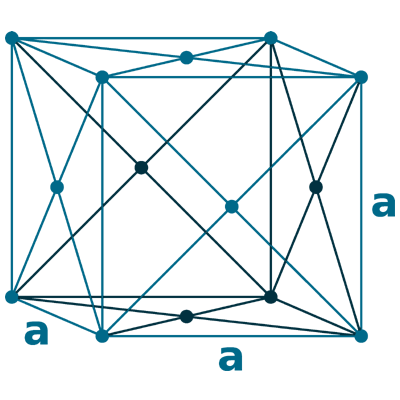
Crystal structure: Face centered cubic
Lattice angles: π/2, π/2, π/2
Lattice constant: 404.97, 404.97, 404.97 pm
Grid parameters: a=4.050 Å
Space Group Name: Fm_3m
Space Group Number: 225
Reactivity of Aluminium
Electronegativity: 1.61 (pauling scale)
Valence: 3
Electron affinity: 42.5 kJ/mol
Nuclear Properties of Aluminium
Half Life: Stable (Infinity)
Lifetime: Stable (Infinity)
Quantum Number: 2P1/2
Neutron cross section (Brans): 0.233
Neutron Mass Absorption: 0.03
Isotopes: 26Al 27Al
| Isotope | Abundance (%) | Atomic Mass g/mol | Half Life (t1/2) |
| 26Al | Trace | – | 7.17×105 y |
| 27Al | 100% | 26.982 | Stable |
Chemical Reactions of Aluminium
Aluminium reacts with air, and forming a protective layer of aluminium (III) oxide, which prevents further reaction with oxygen.
4 Al (s) + 3 O2 (g) → 2 Al2O3 (s)
Due to its protective oxide layer, It doesn’t react with water, But if the oxide layer is damaged, the metal is exposed to attack, even by water.
The metal reacts with all halogens to form aluminium (III) halides:
2 Al (s) + 3 Cl2 (g) → 2 AlCl3 (s) (aluminium (III) chloride)
2 Al (s) + 3 Br2 (g) → Al2Br6 (s) (aluminium (III) Bromide)
2 Al (s) + 3 I2 (g) → Al2I6 (s) (aluminium (III) Iodide)
The Metal dissolves readily in sulphuric acid (H2SO4) or hydrochloric acid (HCl) under the formation of Al (III) & hydrogen (H2), and Concentrated Nitric acid (HNO3) passivates aluminium metal:
2 Al (s) + 3 H2SO4 (aq) → 2 Al3+ (aq) + 2 SO42- (aq) + 3 H2 (g)
2 Al (s) + 6 HCl (aq) → 2 Al3+ (aq) + 6 Cl– (aq) + 3 H2 (g)
Reaction of the metal with Hydroxide ions
Aluminium dissolves in sodium hydroxide (NaOH) with the formation of hydrogen gas (H2), and the formaton of aluminates of the type [Al(OH)4]–:
2 Al (s) + 2 NaOH (aq) + 6 H2O (l) → 2 Na+ (aq) + 2[Al(OH)4]– (aq) + 3 H2 (g)
Aluminium ions are precipitated by Hydroxide (OH–) as a white gelatinous precipitate:
Al3+ (aq) + 3 OH– (aq) ⇌ Al(OH)3 (s)
Aluminium History
Naming: After alumina (aluminium oxide), itself name after mineral Alumen (alum, Latin: meaning bitter salt)
Prediction: Antoine Lavoisier (1782)
Discovery & 1st Isolation: Hnas Christian Ørsted (1824) in Copenhagen (Denmark)
Named by: Humphry Davy (1812)
Aluminium Uses
Aluminium has Low density (light weight), Non-toxicity, durable, excellent corrosion resistance, high thermal conductivity, can be easily cast, machined, & formed, and also Non-magnetic & Non-sparking.
Because of these particular properties, it is used in a huge variety of products including Cans, Window frames, Kitchen utensils, Foils, outside building decoration, Beer kegs, Aeroplane parts and in thousands of industrial applications.
It has Light weight, good conductor of electricity (about 60% that of copper) and cheaper than copper, Because of these properties, it is used in Electrical transmission lines.
Pure aluminum is soft & lacks strength, but when alloyed with small amounts of manganese, copper, silicon, magnesium, or other elements, it remains lightweight but become strong metal.
These alloys are of vital importance in the construction of Modern Aircraft, Rockets, Ships, and other forms of transport.
When aluminium evaporated in a vacuum, it forms a highly reflective coating for both radiant heat & visible light.
This coating soon form a thin layer of the protective oxide and do not decay/deteriorate as do silver coatings.
This coating have many uses, including Decorative paper, Packaging (cans, foils etc..), Toys, and Telescope mirrors, even almost all modern mirrors are made by using a thin reflective coating of aluminum on the back surface of a sheet of float glass.
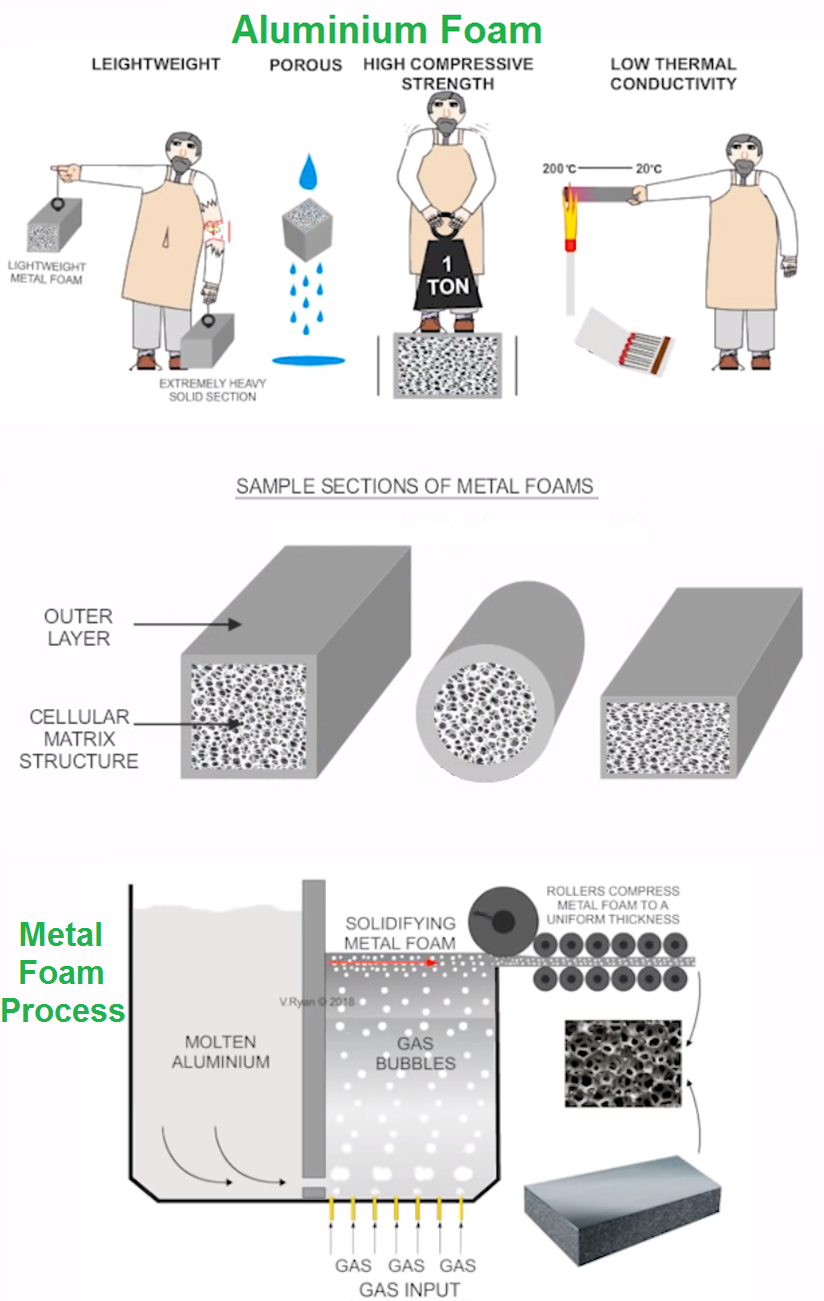
Aluminum Foam (metal foam) is the most recent development in aluminum technology, It produce by adding the molten metal as a compound (a metal hybrid), which releases hydrogen gas.
The molten aluminum has to be thickened before this is done and this is achieved by adding silicon carbide fibers or aluminum oxide.
The result is a solid foam which is used in Space shuttle, Traffic tunnels, and many others applications, where high compressive strength, Impact absorption, low conductivity, vibration absorption are needed.
Biological role of Aluminium
Soluble +3 form of aluminium is toxic to plants.
Acidic soils make up almost half of arable (suitable for growing crops) land on Earth, and the acidity speeds up the release of Al3+ from its minerals, and due to high concentration of Al3+ in groundwater of acidify soil, crops can absorb the Al3+ leading to lower yield or can damage the roots of trees.
Due to Aluminum accumulate in plants, cause health problems for animals that consume these plants.
Human bodies absorbs only a small amount of the aluminium take in with food, but exposed to high concentrations, it can cause health problems, such as Alzheimer’s disease (senile dementia).
Aluminum is a risk in some working environments, such as mines, where it can be found in water can cause lung problems, due to breathe in aluminum dust.
Some foods have more than average concentration of aluminium, Like processed cheese, tea, Lentils, sponge cakes etc..
Cooking normaly in aluminium pans is not harmful, because it does not much increase the concentration of aluminium in our diet, except when cooking acidic foods such as Rhubarb etc…
Abundance of Aluminium
Aluminium is the 3rd (after oxygen and silicon) most abundant metal in the Earth’s crust, but rarely found free in nature.
It is found in minerals such as Bauxite (Al2O3.H2O) & Cryolite (Na3AlF6), these are aluminium silicates.
It is also found in granite & many other common minerals.


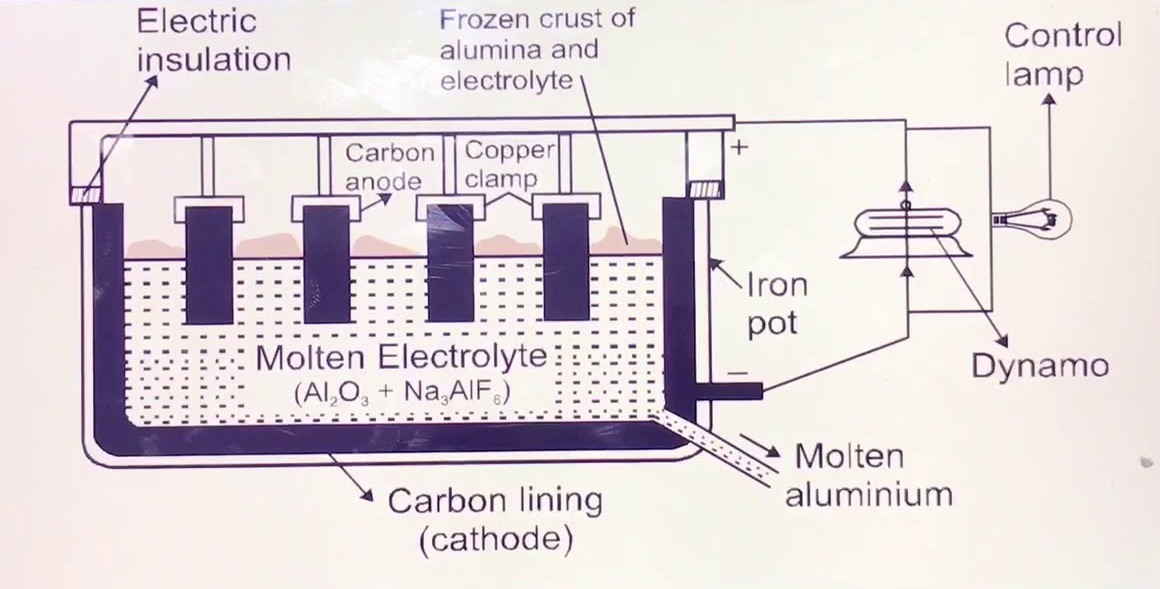
Commercially, Aluminium is extracted by the Hall–Héroult process, In this process pure aluminium is obtained by electrolysis of alumina (aluminium oxide, Al2O3) dissolved in molten cryolite.
This process was discovered by Hall in the U.S and by Heroult in France at the same time in (1886).
For making Aluminium require so much energy, However, once it has been made can be easily recycled, because it does not readily corrode.
5% of the electricity generated is used in aluminium production in the USA.
Aluminum can also be extract from Clay, but at present the process is not economically feasible.
Annual world wide production is around 60,000,000 tonnes.
0.005% (In Universe)
0.9% (In Meteorites)
0.006% (In Sun)
8.1% (In Earth’s Crust)
5×10-7% (In Oceans)
0.00009% (In Humans)
World’s Top producers of Aluminium
1) China
2) Russia
3) Canada
4) India
5) UAE
World’s Top 3 Reserve holders of Aluminium
1) Guinea
2) Australia
3) Brazil
Aluminium Price
Pure Aluminium metal price is around $2.5-$4.5 per KG (KiloGram)
#Aluminium #aluminum


